The Copper Smelting Process
Copper ingots are a refined form of the metal copper, produced through a process called smelting. Smelting involves heating copper ore or scrap copper to high temperatures in a furnace, causing the metal to melt and separate from impurities. The molten copper is then poured into molds to form ingots, which are solid blocks of pure copper.
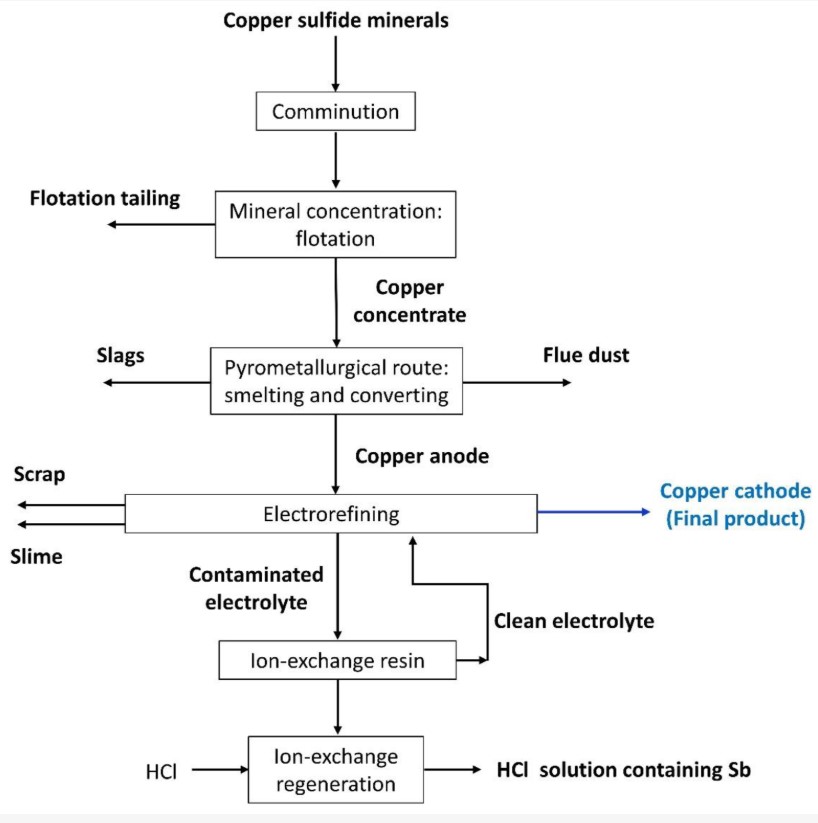
Copper can be produced through two main methods: pyrometallurgy or hydrometallurgy. Pyrometallurgy is a method of converting low-grade copper ore into high-purity metallic copper. This method has three stages:
- Copper processing via flotation
- Converting copper concentrate into metallic copper (copper matte)
- Purification
The hydrometallurgical route is used for a very limited amount of copper resources in the world, and its advantage lies in in-situ leaching of copper ore.
Crushing Stage
The goal of the crushing stage is to reduce the size of the ore particles through crushing, grinding, or other processes, which is an energy-intensive operation with very low efficiency. It is estimated that crushing accounts for 30 to 50 percent of the operating costs of conventional extraction. This will be exacerbated in the coming years due to the lower ore grades, increased rock hardness, and the depth of the mines. Therefore, the industry needs innovative methods to reduce energy consumption in the copper ore crushing process.
- Concentration Stage
The copper concentration stage is usually performed through flotation to separate valuable minerals from gangue materials. The most important factors affecting the performance of the flotation solution are pH. One of the major concerns about flotation is the production of tailings, which still contain copper and other valuable metals. It is expected that tailings production will increase in the coming years due to the intense extraction of low-grade copper ore, and these tailings themselves must be recycled. Therefore, researchers have evaluated methods to improve flotation processes, reduce and reprocess fresh and old tailings.
The pyrometallurgical method, which includes the smelting, converting, and refining stages, aims to separate the metal from the minerals and obtain copper anodes.
- Smelting Stage
This stage differs in converters and rotary furnaces.
–Smelting Stage in Converter Furnaces
The concentrated flotation product (copper concentrate) is converted into sulfide copper matte at a temperature of 1200-1300°C. In the converting stage, air is injected into the liquid phase, which is essentially composed of copper and iron sulfide. The iron sulfide is oxidized, and the copper sulfide is converted into 99% raw molten copper. In the refining stage, the remaining sulfur and oxygen are removed through oxidation.
Finally, the electrolytic refining stage is carried out to purify the copper anode and produce the final copper product as a cathode. One of the main challenges in this process is controlling the concentration of metals in the tank house electrolyte that dissolve from the anode along with the copper, such as arsenic, antimony, and bismuth. Antimony and bismuth are valuable metals widely used in semiconductors, thermoelectrics, pharmaceuticals, chemicals, ceramics, and pigments; they are also considered critical elements by the European Commission. Therefore, the extraction and recovery of these metals from copper production has become increasingly important. In recent years, various methods have been tested to remove these impurities from tank house electrolytes, such as chemical precipitation, solvent extraction using activated carbon, chemical leaching, electrodialysis, and ion-exchange resins. Among these technologies, ion-exchange resins are most widely used on an industrial scale worldwide.
–Smelting Stage in Rotary Furnaces
Sulfide copper concentrate or recycled materials are smelted at 1300-1400°C. The temperature is then reduced, and oxygen is introduced to continue the process until the copper matte reaches a grade of 70% and is separated from the slag. In the refining stage, the sulfide copper matte is oxidized to a grade of 97-99%.